Dr. Hekmat CONSULTING & Interim Management
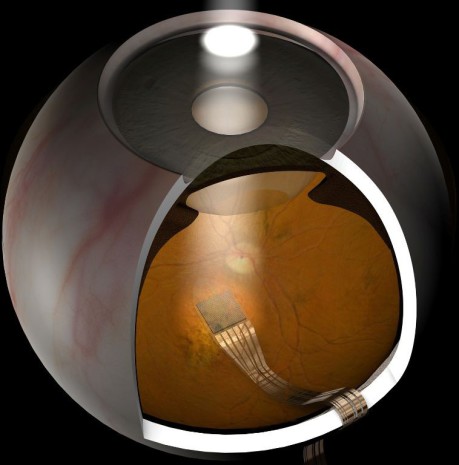
Active Neuro-Implant for Ophthalmology
for partial restoration of vision of the totally blind
Schematics of sub-retinal implant, courtesy of Oxford eye hospital 2013-02-04
Medical diagnostic imaging
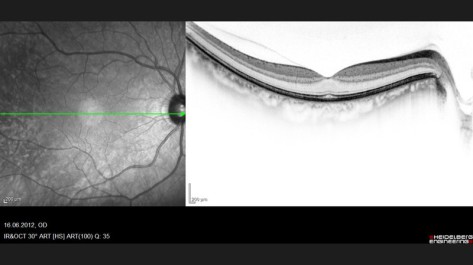
OCT Optical Coherence Tomography
OCT - Dr. A. Hekmat 2012-06
Spectral Domain Optical Coherence Tomography (SD-OCT)
Source: heidelbergengineering.com/international/products/spectralis/technology/spectral-domain-oct/
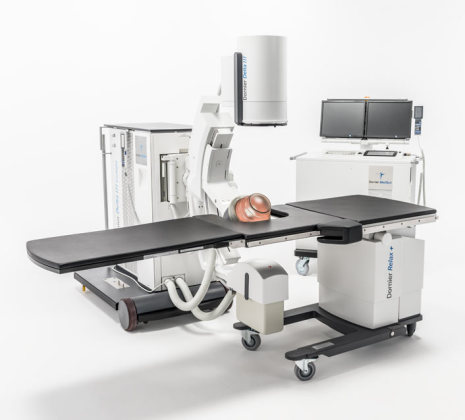
X-ray imaging
via C-arm for targeting of renal stones and lithotripsy
Source: https://www.dornier.com/products/
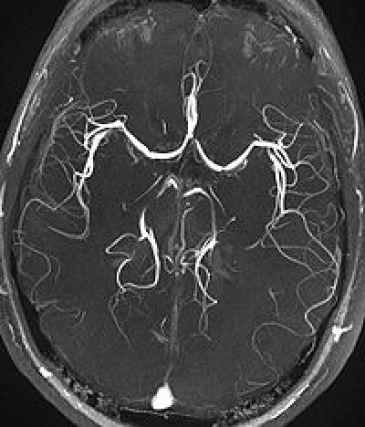
MRI Magnetic Resonance Imaging / Angiography
Contact us to evaluate / validate your Imaging process.
Dr. Hekmat CONSULTING